Is Gabapentin a Controlled Substance? Schedule, State Laws & PDMP Reporting (2026)
Is gabapentin a controlled substance? It depends on the state. This 2026 guide covers gabapentin's schedule, PDMP reporting requirements, state-by-state laws, and what clinicians need to know about prescribing gabapentin safely.
Get a cited, state-specific answer in seconds
Rx Agent gives prescribers and pharmacists instant, sourced answers to pharmacy law and clinical compliance questions.
Is Gabapentin a Controlled Substance in Your State? Use This Tool
It depends on the state. The DEA has not scheduled gabapentin federally — but eight states classify it as Schedule V, more than a dozen require PDMP reporting without scheduling it, and the rest leave it unregulated. Getting this wrong can mean a rejected prescription, a board complaint, or a missed PDMP check.
Is gabapentin a controlled substance in your state?
Select a state to see the current schedule status and PDMP requirement.
Select a state above to check gabapentin's status.
What Schedule Is Gabapentin? Gabapentin Regulatory Map (2026)
I learned that the hard way in 2023 when I wrote a gabapentin refill for a patient who had moved to Virginia — the pharmacy rejected it because I hadn't followed Schedule V prescribing rules. Same drug, same dose, completely different obligations.
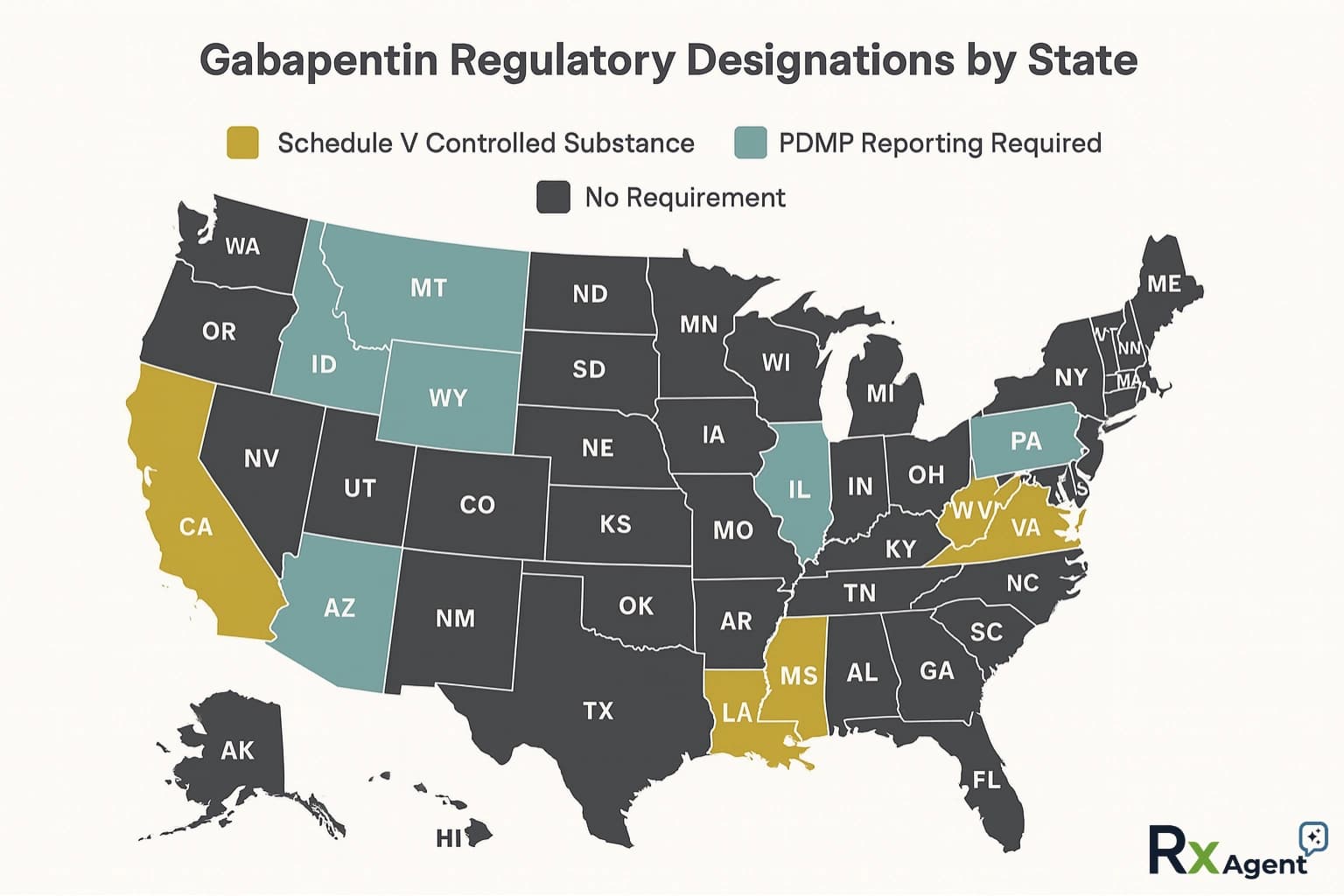
Figure: Current state designations for Schedule V vs PDMP reporting.
Gabapentin Schedule by State (2026)
| Category | States |
|---|---|
| Schedule V + PDMP | AL, KY, ND, TN, VA, WV, UT, MT |
| PDMP Only (not scheduled) | CT, DC, FL, IL, IN, KS, MA, MN, NE, NJ, NM, NC, OH, OR, WI, WY |
| Descheduled (was Schedule V) | MI |
| Under consideration | MD, NY |
| No regulation | All remaining states |
Is Gabapentin a Controlled Substance in Florida?
Gabapentin is not scheduled in Florida, but PDMP reporting is required for every dispensing. Rules may tighten — use the checker above for the current cited statute.
Check Florida gabapentin rules →
Is Gabapentin a Controlled Substance in Texas?
Texas does not schedule gabapentin and has no PDMP reporting requirement — one of the least restrictive states. That can change; verify before prescribing across state lines.
Check Texas gabapentin rules →
Is Gabapentin a Controlled Substance in Indiana?
Indiana does not schedule gabapentin but requires mandatory PDMP reporting via the INSPECT database. Every fill is tracked even without a formal schedule.
Check Indiana gabapentin rules →
Is Gabapentin a Narcotic?
No—gabapentin is not a narcotic, and classifying it as one is a common misconception.
Narcotics refer specifically to opioid drugs like morphine, oxycodone, and fentanyl. Gabapentin is an anticonvulsant (also called an antiepileptic) that works by modulating voltage-gated calcium channels [1]—a completely different mechanism than opioid receptor binding.
So why does the question "is gabapentin a narcotic?" come up so often? In my experience, it's because gabapentin can cause sedation, euphoria at high doses, and physical dependence with withdrawal symptoms—effects that overlap with narcotics in ways patients notice [3]. When gabapentin is misused, especially alongside opioids, it can mimic opioid-like effects. But pharmacologically, gabapentin and narcotics are unrelated.
Is Gabapentin an Opiate?
No—gabapentin is not an opiate or opioid.
Opiates are substances derived from the opium poppy, and opioids are the broader class that includes synthetic versions. Gabapentin belongs to neither category. It does not bind to mu, kappa, or delta opioid receptors [1]. It doesn't produce respiratory depression through opioid pathways, and it's not reversed by naloxone.
The confusion often arises because gabapentin frequently appears in toxicology reports alongside opioids. In 2020, our regional toxicology team found gabapentin in more than 20% of opioid-involved overdoses they reviewed [4] [13]. But appearing together doesn't make them the same drug class. Gabapentin enhances sedation when combined with opioids [2] [5]—which is precisely why states are tightening oversight.
Why Did States Start Making Gabapentin a Controlled Substance?
It wasn't one big event—just a slow, undeniable trend.
Between 2018 and 2023, our clinic increasingly caught diversion cases. In one review, 6 out of 42 high-risk patients had documented gabapentin misuse. Published data shows that misuse rates range from 15–22% among opioid-using populations and up to 40–65% among certain high-risk groups with prescriptions [3]. States responded accordingly: more scheduling, more PDMP reporting, and more oversight [11]. By 2025, more than half the U.S. had some form of gabapentin-specific regulation.
Gabapentin Prescribing Information: What Clinicians Should Know
Beyond scheduling, gabapentin prescribing information matters for safe clinical use.
When prescribing gabapentin, clinicians should keep these essentials in mind:
- FDA-approved indications: Postherpetic neuralgia (adults) and adjunctive therapy for partial-onset seizures (adults and pediatric patients ≥3 years). All other uses are off-label [1].
- Dosing range: Typically 300–3,600 mg/day in divided doses, depending on indication. Dose adjustments are necessary in renal impairment [1].
- Black box warning: None for gabapentin specifically, but the entire anticonvulsant class carries a suicidality warning [1].
- Tapering: Never discontinue abruptly. I taper over at least 7 days—longer for patients on high doses—to avoid withdrawal seizures [1] [12].
- Drug interactions: Enhanced CNS depression when combined with opioids, benzodiazepines, or alcohol. Co-prescribing with opioids requires careful monitoring for respiratory depression [2] [5] [14].
- Telehealth prescribing: If your patient is in a state where gabapentin is a controlled substance, telehealth prescriptions must follow that state's controlled substance rules—not yours. The gabapentin schedule in the patient's state determines your prescribing obligations.
Always verify gabapentin prescribing information against the most current FDA labeling and your state's pharmacy board guidelines.
Frequently Asked Questions
About the Author
Dr. Zade Shammout, PharmD writes about prescription medications, pharmacy laws, and healthcare compliance for prescribers and pharmacists.
