Physician Assistant Prescribing Authority by State (2026)
Updated for 2026. PA prescribing authority across all 50 states — Schedule II–V rules, DEA requirements, supervision, and telemedicine laws. Cited to statute.
Need the exact rule for your state and role?
Rx Agent returns the cited statute, last-reviewed date, and compliance notes for your state, drug schedule, and prescriber type — in seconds.
Can physician assistants prescribe medication?
Yes — in all 50 states. However, Physician Assistant (PA) prescribing authority varies significantly by state, especially for Schedule II controlled substances, telemedicine prescribing, and DEA registration requirements.
This 2026 guide synthesizes those differences, focusing on:
- State-level prescriptive models
- Controlled substance schedules (II–V)
- DEA and telemedicine prescribing rules
- Commonly queried drugs like Wegovy, Adderall, and Norco
- The new DEA Special Registration for telehealth providers
The Legal Landscape: Supervision vs. Collaboration
Most states require a defined relationship between PAs and physicians. This can take one of several forms:
| Model | Description |
|---|---|
| Supervised | PA requires direct or indirect oversight from a licensed physician. |
| Collaborative | PA collaborates with a physician, but operational autonomy is higher. |
| Transition-to-Practice | PAs gain independence after completing a specific number of supervised hours. |
| Independent (OTP) | No legal physician tether; scope is defined at the practice level. |
The Optimal Team Practice (OTP) model, pioneered in North Dakota, eliminates the requirement for a formal supervisory agreement. States like Utah, Wyoming, and Montana are moving in this direction by allowing scope determination at the practice level after 8,000–10,000 hours of experience.
Can Physician Assistants prescribe medication without a doctor?
Generally, no — but the tide is shifting.
Most states require PAs to have a supervision or collaboration agreement with a physician to prescribe. However:
- North Dakota grants full independence under Optimal Team Practice.
- Utah and Wyoming determine scope at the practice level, effectively removing state-mandated supervision ratios.
Compared with nurse practitioners (NPs)—many of whom have achieved Full Practice Authority (FPA)—PAs remain more regulated. The PA advocacy community continues to lobby for expanded OTP legislation that allows flexible, team-based scope determination.
For Nurse Practitioner-specific rules and Full Practice Authority states, see our Nurse Practitioner Prescriptive Authority by State guide.
Common Prescribing Questions: Wegovy, Adderall, Norco & More
Patients and providers often ask: What medications can physician assistants prescribe? Here are common examples that illustrate how laws differ:
Can PAs prescribe Wegovy (Semaglutide)?
✅ Yes. Physician assistants can prescribe GLP-1 receptor agonists like Wegovy and Ozempic in all 50 states, provided they have prescriptive authority and, in some states, a collaborating physician agreement.
Can PAs prescribe Adderall?
⚠️ Usually yes, but Adderall is a Schedule II controlled substance, so prescribing rules are stricter. Some states restrict Schedule II prescribing to specific settings (e.g., hospitals or hospice) or impose day-supply limits.
Can PAs prescribe Norco (Hydrocodone/Acetaminophen)?
✅ Yes, though with Schedule II limits. For example:
- Arkansas & Missouri: Limited to hydrocodone combination products only.
- Florida: May prescribe Schedule II opioids but only for a 7-day supply to opioid-naive patients.
Can PAs prescribe peptides or compounded therapies?
⚠️ Only if the peptide is FDA-approved. Compounded peptides face more scrutiny, and PAs must ensure compliance with both federal FDA and state board rules.
Can a Physician Assistant Prescribe Controlled Substances?
Yes—but only within their state’s statutory limits and with the appropriate DEA registration.
Controlled-substance authority follows a two-tier rule:
- State Law: Defines which schedules (II–V) a PA may prescribe and under what conditions.
- Federal Law (DEA): Requires the PA to obtain a DEA registration and follow national prescribing standards.
For example:
- Georgia prohibits PAs from prescribing any Schedule II drug.
- Texas allows it only in hospital or hospice settings.
- North Dakota permits full Schedule II–V prescribing authority under OTP.
Can Physician Assistants Prescribe Controlled Substances via Telemedicine?
Yes — if BOTH state law and federal DEA telemedicine requirements are satisfied. To prescribe Schedule II–V medications remotely, a PA must:
- Be authorized under state scope-of-practice law
- Hold an active DEA registration
- Comply with the Ryan Haight Act or Special Registration rules
- Conduct PDMP checks where required
- Maintain DEA registration in each state where patients are located
Why It Matters
Under the Ryan Haight Online Pharmacy Act, prescribing controlled substances via telemedicine previously required an in-person evaluation. This was temporarily waived during COVID-19, but those waivers expire at the end of 2025.
The new rule allows compliant PAs to prescribe drugs like Adderall or Testosterone remotely—provided all DEA and state requirements are satisfied.
Physician Assistant (PA) Prescribing Authority by State Table (2026)
The following table synthesizes data regarding the practice environment for PAs, specifically focusing on their ability to prescribe controlled substances (CS).
- Independent: No physician relationship mandated by state law
- Transition: Independence allowed after an hours/experience threshold
- Collaborative: Collaborative agreement required
- Supervised: Supervision required
- Sch II: Schedule II Controlled Substances
| State | PA Authority | Key Prescribing Notes |
|---|---|---|
| AL | Supervised | PA: Sch III-V only. Sch II prohibited [3]. |
| AK | Collaborative | PA: Sch II-V requires MD authorization [3]. |
| AZ | Collaborative | PA: Transition to independent after 8k hours [3]. |
| AR | Supervised | Restriction: Sch II limited to hydrocodone products only [3]. |
| CA | Supervised | |
| CO | Collaborative | PA: Sch II limited to 7-day supply for opioid naive [3]. |
| CT | Supervised | PA: Sch II/III req MD approval [3]. |
| DE | Collaborative | PA: Sch II-V authorized [3]. |
| FL | Supervised | PA: Sch II limited to 7-day supply [3]. |
| GA | Supervised | Restriction: PA prohibited from prescribing Sch II [3]. |
| HI | Supervised | PA: Sch II-V under MD supervision [3]. |
| ID | Collaborative | PA: No written or oral prescriptions allowed [3]. |
| IL | Collaborative | |
| IN | Collaborative | PA: Sch II-V requires agreement. No weight loss Sch IIs [3]. |
| IA | Supervised | PA: Sch II depressants req MD approval [3]. |
| KS | Supervised | PA: Sch III-V only [3]. |
| KY | Supervised | |
| LA | Supervised | |
| ME | Collaborative | PA: Independent after 4k hours [3]. |
| MD | Collaborative | |
| MA | Supervised | PA: Sch II requires 96hr MD review [3]. |
| MI | Collaborative | PA: Practice agreement required [3]. |
| MN | Transition | |
| MS | Supervised | |
| MO | Collaborative | Restriction: Sch II limited to hydrocodone. 5-day limit [3]. |
| MT | Transition | PA: Independent after 8k hours [3]. |
| NE | Supervised | |
| NV | Supervised | PA: Sch II-V authorized [3]. |
| NH | Transition | PA: Independent after 8k hours [3]. |
| NJ | Supervised | PA: Sch II-V authorized [3]. |
| NM | Collaborative | PA: Sch II-V authorized [3]. |
| NY | Supervised | |
| NC | Supervised | PA: Sch II/III limited to 30-day supply [3]. |
| ND | Independent | Standout: PA practices independently [3]. |
| OH | Supervised | PA: Sch II restrictions (e.g., terminal illness, MD initiation) [3]. |
| OK | Supervised | Restriction: PA excluded from independent Sch II [3]. |
| OR | Collaborative | PA: Sch II-V authorized [3]. |
| PA | Supervised | PA: Sch II limited to 72hr (initial) or 30-day (ongoing) [3]. |
| RI | Collaborative | PA: Sch II-V authorized [3]. |
| SC | Supervised | Restriction: PA Sch II limited (5-day narcotic/30-day non) [3], [4]. |
| SD | Collaborative | |
| TN | Collaborative | PA: Sch II/III limited to 30-day supply [3]. |
| TX | Supervised | Restriction: Sch II limited to hospital/hospice settings [3]. |
| UT | Transition | PA: Independent after 10k hours [3]. |
| VT | Collaborative | PA: Sch II-V authorized [3]. |
| VA | Collaborative | |
| WA | Transition | PA: Independent after 4k hours [3]. |
| WV | Collaborative | PA: Sch II limited to 3-day supply [3]. |
| WI | Collaborative | |
| WY | Collaborative | PA: Sch II-V authorized [3]. |
PA Prescribing Authority by State Map (2026)
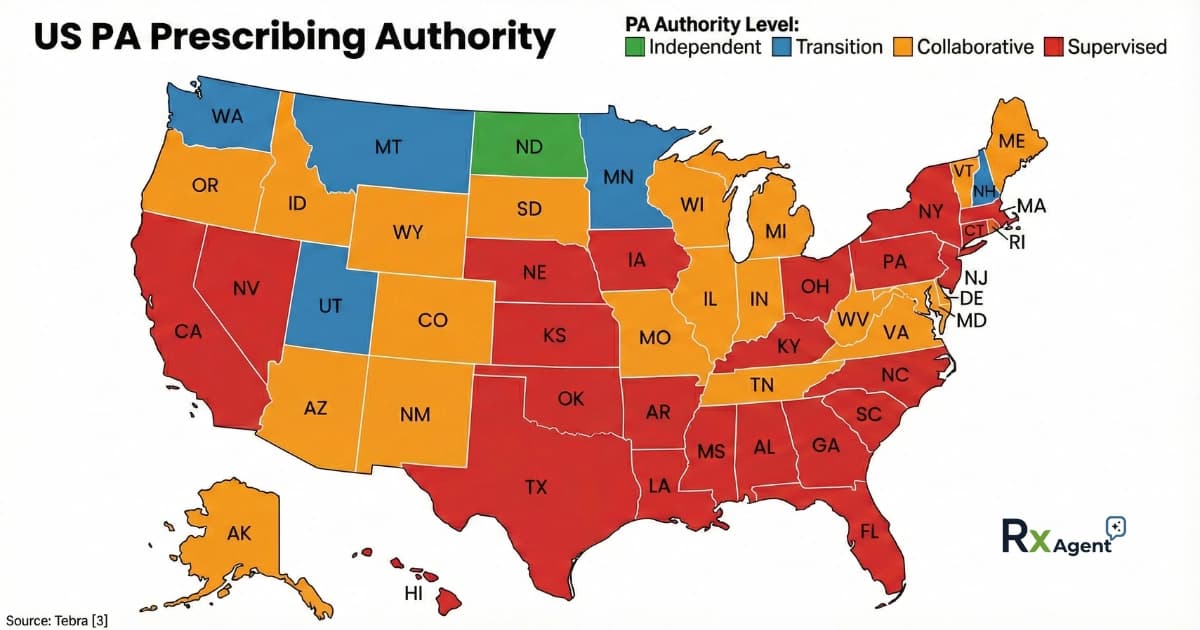
Figure: State-by-state breakdown of Physician Assistant prescribing authority, showing independent, transition, collaborative, and supervised practice environments.
Why Manual Compliance Checks Fail
Because physician assistant prescribing authority varies by state — and changes frequently — manual spreadsheets and static PDF charts become outdated quickly.
For Example:
- A state may allow Schedule II prescribing in hospitals but not outpatient clinics
- Telemedicine exceptions may change annually
- PDMP documentation requirements vary by state
Meet Rx Agent — Your AI Compliance Tool
Rx Agent is the best telehealth compliance software designed for prescribers and telemedicine organizations.
It acts as your automated compliance officer, helping you:
- Instantly verify state-specific prescriptive authority
- Cross-reference DEA and telemedicine laws
- Get real-time updates when regulations change
- Eliminate manual cross-checking and compliance errors
👉 Start Free Trial and simplify your multi-state prescribing compliance today.
Conclusion: The Future of PA Prescribing Authority
The landscape of physician assistant prescriptive authority by state is rapidly evolving toward greater autonomy.
As Optimal Team Practice expands and DEA telemedicine frameworks mature, PAs are gaining more flexibility—paired with higher compliance accountability.
The key takeaway for PAs and telemedicine providers:
✅ Stay informed, stay compliant, and use automated tools, like Rx Agent, to navigate state prescribing laws.
References
1. https://www.aanp.org/advocacy/advocacy-resource/policy-briefs/issues-full-practice-brief
2. https://nurse.org/education/np-full-practice-authority/
3. https://www.tebra.com/theintake/checklists-and-guides/legal-and-compliance/nurse-practitioner-laws-by-state
4. https://www.scstatehouse.gov/billsearch.php?billnumbers=3580
5. https://nacns.org/wp-content/uploads/2020/08/PractPrescAuthority7.31.2020.pdf
6. https://www.dlapiper.com/insights/publications/2025/01/dea-and-hhs-publish-rules-for-telemedicine-prescriptions
7. https://www.govinfo.gov/content/pkg/FR-2025-01-17/pdf/2025-01099.pdf
8. https://www.ncsl.org/scope-of-practice-policy/practitioners/physician-assistants/physician-assistant-practice-and-prescriptive-authority
9. https://www.revisor.mn.gov/statutes/cite/147A.185
10. https://ndlegis.gov/cencode/t43c17.pdf
11. https://www.leg.state.fl.us/statutes/index.cfm?App_mode=Display_Statute&URL=0400-0499%2F0458%2FSections%2F0458.347.html
12. https://www.nysspa.org/page/NYFAQ
13. https://www.govinfo.gov/app/details/FR-2025-01-17/2025-01099
14. https://www.federalregister.gov/documents/2025/01/17/2025-01099/special-registrations-for-telemedicine-and-limited-state-telemedicine-registrations
Frequently Asked Questions
About the Author
Dr. Zade Shammout, PharmD writes about prescription medications, pharmacy laws, and healthcare compliance for prescribers and pharmacists.
